On Google Alert recently there was notice that the Danish Crown has recalled a batch of pork due to traces of the banned hormone, DES. The report details show this product listed for distribution to Australia. Inquiries to the Department of Agriculture by DES Action NSW have revealed this consignment of pork will arrive in Australia in several weeks time.
In response to the European Commission's warning of the goods in transit to Australia, the Food Standards Australia New Zealand (FSANZ) has notified state and territory health authorities to ensure the matter is addressed. It is of concern to DES Action NSW that FSANZ has advised Queensland Health that the pork's concentration of DES is of minimal toxicological concern for humans. Where DES Action NSW's understanding is that there is no known safe level of DES, it is a matter of concern that the Australian response may be based on inaccurate information of there being a safe level.
Today DES Action NSW has written to Minister for Agriculture, Barnaby Joyce MP, stating the absence of scientific evidence of a safe level of DES. The organisation has forwarded the Minister details of research showing DES residue at low doses in agricultural products to be more effective at inducing cancer than large doses, research showing DES residue in beef to alter testicular development and reproductive capacity, and reports of precocious puberty in young children who have ingested DES contaminated food. In view of this evidence provided, the Minister is urged to comply with zero tolerance for DES residue in imported food as per the Australian Policy Guideline on the Regulation of Residues. Request has been made for border action under the Imported Food Inspection Scheme to assist State government authorities deal with this matter, or even adopting stricter measures.
Recent media report Local Pork industry calls for Australian-made bacon labelling overhaul -Landline 27/7/14 raises concern that imported Danish pork is an accepted ingredient in packaged bacon labelled as an Australian product. Department failure to address DES contaminated pork could mean the spread of DES into Australian bacon products.
Sunday 10 August 2014
Thursday 29 May 2014
Hunt for a DES drug Biomarker gives hope for
DES exposed Australians
US researchers from the National Cancer Institute are on the hunt for a biomarker to prove prenatal exposure to the anti-miscarriage drug DES (diethylstilboestrol). Using advanced scientific tools and blood donated by recruits from the National Cancer Institute's DES Follow-up Study, it is hoped a biomarker will be discovered by early next year to identify people exposed to DES in the womb.
"A simple blood test to prove DES exposure in the womb would help many Australians previously uncertain of DES exposure to determine their need for special health care," says Carol Devine, coordinator of the support advocacy group DES Action NSW.
"So far successive governments have refused to promote the DES topic directly to the Australian public in health promotion programs due to concern about raising anxiety in people uncertain of DES exposure."
"But this has meant neglecting the rights of all Australians to be informed of the possibility of DES exposure, with the majority of DES exposed people suffering DES effects without knowing why and what they can do about it," adds Devine.
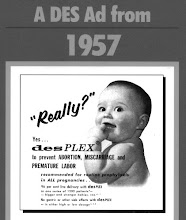
DES was prescribed worldwide to over ten million pregnant women over three decades to prevent miscarriage. In the early 1970s it was linked to clear cell adenocarcinoma of the vagina/cervix in women exposed to DES in the womb. It is now known to cause reproductive problems in DES-exposed offspring and increased risk of breast cancer in women given DES during pregnancy and their daughters of that pregnancy. People exposed to DES during pregnancy and in the womb require lifelong specialised vital health care to help detect DES associated cancers early. DES Action NSW estimates about 740,000 Australians have been exposed to DES.
For DES Awareness Week June 1-7, as well as assisting people learning about their DES exposure for the first time, the DES Action group will be spreading the hopeful news of the DES drug biomarker research. During the Awareness week, June 7 will be set aside to reflect on those who have lost their lives due to DES exposure.
More information about DES can be found at DES Action NSW website: www.desnsw.blogspot.com and on the Cancer Council website at: http://www.cancercouncil.com.au/880/news-media/get-the-facts/cancer-prevention-get-the-facts/cancer-council-new-south-wales-diethylstilbestrol-des-and-cancer/ or by phoning DES Action NSW 02 98754820.
-END-
Information sources:
http://us5.campaign-archive2.com/?u=317818521a5c55131fb1beaf1&id=33b47bfd59&e=a002bf1fe2
http://dceg.cancer.gov/about/staff-directory/biographies/O-Z/hoover-robert
http://www.desfollowupstudy.org/index.asp
Correspondence: Dr Michael Wooldridge 12/10/99, Minister for Health and aged Care (regarding community anxiety with DES exposure information)
For more information or interviews, contact:
Carol Devine
E: c_devine@bigpond.net.au
P: 02 98754820
Wednesday 9 April 2014
9th April 2014
Indian website promotes dangerous DES drug
Shock waves were sent through the Australian group, DES Action NSW last week when its members discovered that DES (diethylstilboestrol) is currently listed for usage in pregnancy on the Indian website Medindia. This discovery supports what the DES Action group has both feared and suspected about this cancer-causing drug since receiving anecdotal stories in the 1990s of its continued usage in pregnancy after it having been contraindicated from this usage some twenty years earlier.
DES was prescribed worldwide to over ten million pregnant women over three decades to prevent miscarriage. In the early 1970s it was linked to clear cell adenocarcinoma of the vagina/cervix in women exposed to DES in the womb. It is now known to cause reproductive problems in DES-exposed offspring and increased risk of breast cancer in women given DES during pregnancy and their daughters of that pregnancy. People exposed to DES during pregnancy and in the womb require lifelong specialised health care.
"In the 1990s we received word from some south-western Sydney doctors and doctors from overseas studying at University of NSW that DES was being prescribed in pregnancy in Pakistan, Bangladesh, and Sri Lanka," says Carol Devine, DES Action NSW coordinator. "No words can describe the shock these doctors felt when learning from us for the first time about harm caused by DES, whilst having knowledge that DES was being prescribed during pregnancy in their respective home countries," says Devine.
Medical experts at a 1992 symposium in Sweden made calls for measures to address the continued usage of DES in pregnancy in many parts of the world, including East and Central Europe, Africa, Asia and Latin America. However, it is unclear what actions ensued. The DES Action NSW group is now urging women who may have been given DES during pregnancy in India or any doctors with knowledge of this usage to report this to government medical authorities in India.
The DES Action group is concerned that there may even be women or doctors from India now living in Australia who have information on this to report. The group is interested in hearing from these people.
"The apparent inertia in dealing with continued DES usage is tragic, and in retrospect it should have always been incumbent on governments worldwide to issue frequent warnings about the danger of DES," comments Devine.
More information about DES can be found at Cancer Council NSW and the contact for DES Action NSW is 02 9875 4820/c_devine@bigpond.net.au
-ENDS-
References:
http://www.medindia.net/drugs/medical-condition/Miscarriage.htm (DES indicated for usage during pregnancy to prevent miscarriage)
http://www.medindia.net/doctors/drug_information/diethylstilbestrol.htm (DES usage is indicated in pregnancy and then its listing as contraindicated creates confusion)
http://desnsw.blogspot.com.au/2009/04/real-multicultural-affair.html
Subscribe to:
Posts (Atom)