WHAT I THOUGHT I KNEW
WHAT I THOUGHT I KNEW Alice Eve Cohen ISBN 978-0-670-02095-9
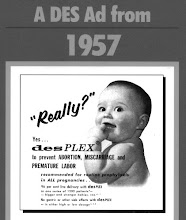
Solo theater artist Alice Eve Cohen knew that childbearing was simply impossible—her own mother had taken DES, and Alice had a deformed uterus, among other disqualifiers. So when what doctors misdiagnosed as a tumor turned out to be a 6-month fetus, the 44-year-old Cohen had to wrestle with clueless specialists, cavalier insurance companies, and her own no-see-um maternal instincts. Her darkly hilarious memoir, What I Thought I Knew (Viking), is an unexpected bundle of joy.