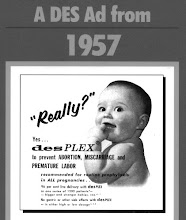
A pilot project is currently being undertaken by DES Action Australia-NSW to determine whether targeting pharmacy customers is the most effective means of raising DES awareness in the Australian community. This involves interested retail pharmacies displaying DES information for a 12 month period (if possible).
....and now we need your help
If you think your local pharmacy or pharmacies might be interested, please contact 02 98754820 or c_devine@bigpond.net.au for a Pharmacy DES information kit for you to take along to the pharmacy and then confirm interest at the above contact. Thank you!
Thursday, 13 December 2012
Sunday, 21 October 2012
DES victims call to sisterhood
Over four decades the great majority of an estimated 740,000 women prescribed DES during pregnancy and their children exposed to DES in the womb have been unaware of the danger of DES. Consequently they have been missing out on information about the vital special health care for their increased risks of clear cell adenocarcinoma of the vagina/cervix (CCA), breast cancer and other reproductive problems.
"A better understanding of the DES health condition and how this issue came to fall off the radar of health authorities and medical professionals will help major women's groups realise the importance of addressing the problems experienced by the Australian DES exposed population, the majority of whom are women," says group coordinator, Carol Devine.
It is common for the group to receive phone calls from women who have suffered the effects of DES without knowing why, and are only now learning about the harm caused by DES and the rightful health care they need through the group's own media efforts. Last year the New England Journal of Medicine revealed women exposed to DES in utero are at an increased risk of at least 12 adverse health conditions - in some cases up to three times that of other women.
Researchers are still following the health of the DES exposed population to determine whether other health problems occur with age and whether subsequent generations are affected. Women's groups interested in information can contact DES Action Australia-NSW, c_devine@bigpond.net.au / 02 9875 4820.
From National Cancer Institute USA.
Saturday, 4 August 2012
WONDER DRUG almost makes it!
This week the film in development, WONDER DRUG was so very close to winning public votes in a survey by Indiewire. The winning margin was only 3%!!
You can see more about WONDER DRUG at http://www.wonderdrugthemovie.com/
Thank you to everyone who voted. We are all supportive of screen writer and DES daughter, Caitlin McCarthy in her quest for Wonder DRUG to make it to the big screen. With this, there is hope the film will be shown Australia and further raise consciousness of drug disasters. And yes, drug disasters look set to continue with the now usage of dexamethasone in pregnant women to prevent intersex, tomboys and lesbians ( http://www.northwestern.edu/newscenter/stories/2012/08/dreger-fetal-engineering.html ). This report shows NOTHING has been learned from the horrors of the DES and thalidomide experience - thus making WONDER DRUG all the more important for viewing audiences world-wide.
You can see more about WONDER DRUG at http://www.wonderdrugthemovie.com/
Thank you to everyone who voted. We are all supportive of screen writer and DES daughter, Caitlin McCarthy in her quest for Wonder DRUG to make it to the big screen. With this, there is hope the film will be shown Australia and further raise consciousness of drug disasters. And yes, drug disasters look set to continue with the now usage of dexamethasone in pregnant women to prevent intersex, tomboys and lesbians ( http://www.northwestern.edu/newscenter/stories/2012/08/dreger-fetal-engineering.html ). This report shows NOTHING has been learned from the horrors of the DES and thalidomide experience - thus making WONDER DRUG all the more important for viewing audiences world-wide.
Wednesday, 23 May 2012
DES Cancer Cases Go Astray From TGA's Watch
DES daughters diagnosed with clear cell adenocarcinoma of the cervix/vagina (CCA) are urged to self-report their cases to the Adverse Medicines Events Line Ph 1300 134237. Also, to obtain forms for self-reporting to the International DES Cancer Registry, call DES Action Australia-NSW Ph 02 98754820. It doesn't matter if you think a health care provider may have already reported your case, because all reports are cross-checked.
There are many important reasons for self-reporting:
- Case reports are crucial to knowing the magnitude of the DES exposure problem in our country.
- DES Action Australia-NSW knows of instances where cases have gone unreported by health care providers in Australia.
- The Therapeutic Goods Administration (TGA), Australia's drug watchdog, deemed DES cancer reports unnecessary from 1983 to 2001.
- In TGA documents obtained via FOI by DES Action Australia-NSW there was mention of at least 12 cases of CCA, but these were not reported/investigated for entry into TGA data. Of these cases, one woman had died and at least 5 were children.
Friday, 11 May 2012
PIP Inquiry Rejects DES Story
Here are some excerpts from the 9 page submission:
"The monitoring and follow-up of the DES drug disaster by the TGA has been found wanting, with its initial delay in response and inefficiencies in administering DES cancer reports, sometimes with practices that would appear unorthodox (ie, the non-acceptance of data from 1983-2001). Lack of interest in investigating and acceptance of its own flawed and incomplete data has meant no further concerted action in continuous monitoring of the DES problem both in Australia and overseas by the TGA. Even where possibilities have existed to improve its own database, the TGA asserts there are restrictions in its role, eg, it is not in its role to cross-check the International DES cancer Registry. There is no feasible explanation for the misleading and inaccurate media release that was issued to the public (and for the government) in 2004. The reliability of its information sources for the 2004 media release should be questioned, including its available resource, RANZCOG, with its specific representative for DES health matters. The TGA's media release in 2004 provides evidence of failure to consult with USA experts in the DES field of research, some of whom are world renowned. Therefore, the TGA's procedures in risk assessment of DES exposed Australians could not be considered comprehensive and effective when only Australian consultative bodies have been used. If the TGA has any procedures in place to communicate with general public in relation to DES, then these have ensured that the Australian public has remained uninformed, ill-informed or has been issued accurate information ineffectively.
In summary, by its actions and inactions, the TGA has stymied the availability of data to the Department of Health and Ageing in relation to the magnitude of the DES problem in Australia. By its actions and inactions, the TGA has stymied the likelihood of the Australian public receiving accurate information about DES. The TGA's response to the DES exposure problem has had an overall effect of decreasing government attention to the DES exposure problem - to the detriment of DES exposed Australians."
"The TGA response to the PIP disaster has been similar to the DES disaster response with self-imposed limitations in the acquisition of risk-assessment information. There has been limited focus on activities by overseas regulators and limited access to available international data in relation to PIP implant rupture rates. Similar to the DES problem, unbalanced and poor quality information to the government has resulted in sub-standard assistance to PIP breast implant recipients. Once again, the TGA's advice regarding a dangerous "therapeutic" good is grossly inadequate and not in line with overseas regulatory bodies and governments. Appropriate government response has been further impeded where there are ongoing problems, eg, surgery difficulties and arising complications when removing ruptured implants, substance leakage even in implants not ruptured. Just as PIP breast implant recipients have been deserted by the TGA and government with "time bombs" in their bodies, DES exposed Australians have similarly been disenfranchised with the "time bomb" effect of DES. Indeed, there may be DES daughters who have succumbed to DES related breast cancer and subsequently become PIP breast implant recipients. These women would have in effect, fallen foul to TGA inefficiency twice over."
"The fact that the TGA is fully funded by fees from the pharmaceutical industry provides explanation for its poor and inhumane responses to serious drug/medical device adverse events. This very obvious conflict of interest and the increase in reported incidents should signal the need for government legislation to ensure that specialist task forces independent of the TGA (with role of consulting with affected stakeholders), are set up to respond to medical device/drug disasters."
Without due consideration of this submission as part of this Inquiry, the shortfalls in TGA responses to any drug/medical device disaster will not be recognised. This is tragic for PIP recipients and all other victims of drug/medical device disasters.
Friday, 13 April 2012
Global DES Health Survey

DES Action USA with a researcher has now created an online DES Health History Survey targeting DES exposed individuals world-wide.
That's right! This includes Australians!
This survey will help identify health issues faced by women who took DES, their DES exposed children, and also their children (DES granddaughters and grandsons). Hopefully, DES Action USA will then be able to share information of any health trends with researchers to enable their follow-up with further study. The survey is not a formal scientific study, and so there is not the usual informed consent process with privacy information.
The deadline for the completion and return of this survey is JUNE 15th 2012.
To get to the survey go to : http://desaction.org/
Please share news of this survey with any friends or relatives you know to have been exposed to DES.
Let all of our health experience be counted - from everywhere in the world!
Sunday, 22 January 2012
MEDIA RIPPLE EFFECT NEEDED - from UK to OZ
 The UK newspaper The Independent (front page) today has raised awareness of the DES exposure problem for the benefit of UK's population. See:
The UK newspaper The Independent (front page) today has raised awareness of the DES exposure problem for the benefit of UK's population. See:http://www.independent.co.uk/life-style/health-and-families/health-news/thousands-of-women-could-be-at-risk-from-silent-thalidomide-6292889.html
This comprehensive article will no doubt encourage many DES-affected in UK, who have so far been unaware of the harm of DES, to seek information in helping prevent unfavourable health outcomes due to DES.
This article also highlights the latest US Court victory for DES daughters diagnosed with breast cancer and the possible legal opportunities for compensation by UK women so affected. Pleasingly, the article states that US DES lawyer, Aaron Levine, will travel to the UK in two weeks time to coordinate a hunt for the "DES daughters" in UK who have been unable to get compensation in British courts. There has been UK media follow-up on this:
http://topnews.us/content/245785-lawyer-hunt-des-daughters-uk
This interest by UK media has been sparked by the great work of Domino and supportive partner (Diethylstilbestrol, Journal of a DES Daughter). A ripple effect to reach and activate Australian media is needed...followed by the Australian "hunt for DES daughters".
NOTE: DES Action Australia-NSW appeals to all DES affected Australians to contact Australian media, newspapers, radio etc right now to tell them about the attention in US and UK, and the US court victory for DES daughters diagnosed with breast cancer.
Friday, 20 January 2012
Media Release: DRUG COMPANIES ORDERED TO NEGOTIATE SETTLEMENTS IN BREAST CANCER CASE
 Fourteen drug companies in the US have been ordered to begin mediation immediately to compensate 53 women who claim their breast cancers were caused by the anti-miscarriage drug diethylstilboestrol (DES). There are an estimated 740,000 Australian mothers, daughters and sons affected by the drug, which has been found to double a DES daughter's chance of developing breast cancer.
Fourteen drug companies in the US have been ordered to begin mediation immediately to compensate 53 women who claim their breast cancers were caused by the anti-miscarriage drug diethylstilboestrol (DES). There are an estimated 740,000 Australian mothers, daughters and sons affected by the drug, which has been found to double a DES daughter's chance of developing breast cancer.DES was manufactured and sold primarily from 1940 to 1971. It has been linked to rare reproductive cancers, infertility and breast cancer in the daughters born to women who were prescribed it. It was made by numerous pharmaceutical firms, including Eli Lilly and Company, which was the largest producer, and E.R. Squibb and Sons, the predecessor to Bristol-Myers Squibb.
Carol Devine, coordinator of DES Action group, says this case sets a precedent for people affected by DES in the US and around the world. "We are often asked about legal compensation on DES matters, so this is heartening news for Australian DES daughters who have had breast cancer diagnosis," she says.
"There has already been successful litigation in 2008 by an Australian DES daughter in US court against DES drug companies for her diagnosed clear cell adenocarcinoma which left her infertile after a radical hysterectomy."
In agreement with DES Action USA and the College Statement by the Royal Australian and New Zealand College of Obstetricians and Gynaecologists, Carol Devine urges DES daughters to be aware of their increased breast cancer risk once they hit forty and to have annual breast screening.
"This ruling against the drug companies is a wake-up call for the government to promote DES information directly to Australians in public health programs, especially since the majority of DES-affected are still unaware of DES and the associated health care they need."
Jackie White, of Centerburg, OH, is one of the plaintiffs in the case. She, like the other so-called DES daughters, was exposed to DES before birth. "I am just over the moon at this decision," she says. "It is time for the drug companies who harmed us to be held accountable for this bad drug."
Washington DC Attorney Aaron Levine called upon a team of expert witnesses including the Chair of Harvard's Department of Epidemiology, and other scientists from academic institutions such as Georgetown University and the University of Paris to support the reliability of their experts and their opinions that prenatal DES exposure substantially increases the risk of breast cancer in DES daughters over the age of forty.
For more information about DES contact DES Action NSW by phoning 02 98754820 or visiting www.desnsw.blogspot.com
-End-
For more information contact:
Carol Devine, DES Action NSW
E: c_devine@bigpond.net.au
P: (02) 98754820
Tuesday, 3 January 2012
Our DES Awareness Week - a response
 The Daily Telegraph published a controversial article about Events Calendars by Melissa Matheson last week, "What say we call it a day" (31/12/11) with the subtitled comment - "The calendar is now jam-packed with worthy and not so worthy causes - and there's a ribbon for every one."
The Daily Telegraph published a controversial article about Events Calendars by Melissa Matheson last week, "What say we call it a day" (31/12/11) with the subtitled comment - "The calendar is now jam-packed with worthy and not so worthy causes - and there's a ribbon for every one."It was a shame that our DES Awareness Week June 1-7 did not even rate a mention, as this would have fulfilled our whole aim - to raise awareness of DES. Had it been mentioned, it would have certainly been categorised as a worthy cause. However, the fact it did not rate a mention may well demonstrate the stiff competition DES Action NSW as a marginalised unfunded group is up against with other campaigners, armed to the teeth with expensive PR company back-up.
The article had a sweeping statement that campaigners want your attention and, naturally, money. Our campaign has never asked people for money, but instead directly offers Australians information to help prevent cancer fatalities and reproductive health complications - vital health information as a result of a drug disaster that the government currently refuses to provide Australians in public health programs. With the sheer number of calendar campaigns all clamouring for attention, Matheson raises the issue of public exhaustion trying to keep up and the risk that public empathy may turn to apathy. It is hoped this never happens. The response to DES Awareness campaigns over the last seven years has shown the Australian public to be extremely appreciative of the information offered - information they would not have otherwise found.
Events Calendars open a window of excellent opportunity for marginalised organisations with worthy causes. In fair play, media should be focusing equally on those campaigners doing it tough with worthwhile causes. Indeed, a follow-up article about the experience of campaigners, like DES Action NSW, doing the hard grind without any ribbon and stifled by the PR power of larger campaigners, would be a positive move for public good. Focusing on the worthy campaigns at the fringe would bring something new to many people and simultaneously do justice to the underlying principles of Events Calendars.
Subscribe to:
Posts (Atom)